Quantum Dots

Quantum Dots (QD) are semiconductor nanoparticles possessing unique size-dependent optical properties due to quantum confinement. Due to size-tunable bandgap, QDs with luminescence at different wavelengths can be produced simply by changing the crystal size of the QD. Luminescence can be tailored for the visible or near-infrared region by tailoring the composition of QDs.The most studied QDs are the Cd-chalcogenides which have strong absorbance in the UV and emission in the visible region. However, lately, requirements in the medicine and energy areas make the QDs with strong absorbance in the visible and luminescence in the NIR very popular.
In our group, we focus on the synthesis of highly stable colloidal quantum dots emitting in the visible (Cd-chalcogenides) and NIR regions (Pb-chalcogenides and Ag-chalcogenides) and investigate their potential as theranostic nanomaterials, sensors, barcode materials and sensitizers in solar cells.
We are currently working on projects wherein the photothermal therapy (PTT) potential of Ag2S is exploited along with chemotherapy or PDT combinations to achieve tumour-specific image-guided phototherapy or photo/chemotherapy combinations.
https://pubs.rsc.org/en/content/articlehtml/2020/nj/c9nj04608a
https://pubs.rsc.org/en/content/articlepdf/2021/nr/d1nr03507j
https://www.sciencedirect.com/science/article/pii/S1011134420305327
Highlight from our previous QD studies:
Academic and industrial partnership
Our studies on the development of stable, low-cost PbS initiated the establishment of QUANTAG, A.Ş., but also became an enabler for the development of new QD sensitized solar cells.
https://doi.org/10.1021/acsami.0c06694
Towards better luminescence and stability with lower toxicity: Thioacid but which one?
It is well known that the passivization of the QD surface is critical to toxicity, stability, and the quantum yield. Usually, thioacids, such as thioglycolic acid or 3-mercaptopropionic acid, is used for aqueous colloidal QDs. We have discovered that 2-mercaptopropionic acid (2MPA) is a much better thioacid coating in the stabilization of QDs due to possible back binding of the carboxylates on the crystal surface, providing dramatically better luminescence intensity and stability with lower cytotoxicity.
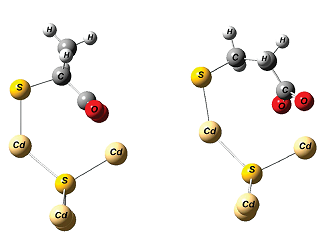
H.Yagci Acar, R. Kas, E. Yurtsever, C. Ozen, I. Lieberwirth, “Emergence of 2MPA as an Effective Coating for Highly Stable and Luminescent Quantum Dots” J Phys Chem Part C, 113, 10005–10012, 2009.
Synergy originating from the mixture of “small“ and “macro” molecules
Cytocompatibility, long term stability, and high quantum efficiency are usually difficult to achieve with QDs. Polymeric coatings, such as poly(acrylic acid) provide long term stability, thioacids provide strong luminescence. We have discovered that the combination of the two can create synergy.

Picture of the luminescent a) CdS-PAA, b) CdS-PAA/MAA 80/20, c) CdS-MAA QDs prepared at 60°C. Samples were excited with a UV lamp at 360nm.
H.Yagci Acar, S. Celebi, N. I. Serttunali, I. Lieberwirth, “Development of Highly Stable and Luminescent Aqueous CdS Quantum Dots with Poly(acrylic acid)/Mercaptoacetic Binary Coating System”, J. Nanoscience and Nanotechnology 2009, 9, 2820–2829.
The emergence of DMSA (2,3-dimercaptosuccinic acid) as a new safe coating and S-source
DMSA is an FDA approved drug for heavy metal poisoning. In order to decrease the toxicity originating from cadmium, its antidote may be used as the coating of a Cd-chalcogenide ! Our studies have shown that DMSA is not only a good coating for QDs but also a slow-S releasing agent which outperforms thioacetamide, the most widely used slow S-releasing agent.

CdS-DMSA quantum dots synthesized by Esra Sevinc.
Sevinc, F. S. Ertas, G. Ulusoy, C. Ozen, H. Yagci Acar, “Meso-2,3-dimercaptosuccinic acid: from heavy metal chelation to CdS quantum dots”, J Materials Chemistry, 22, 5137, 2012.